|
Read more about how to correctly acknowledge RSC content. Permission is not required) please go to the Copyright Proteins such as keratin and collagen can act as moisturizers and also serve as protectors and. If you want to reproduce the wholeĪrticle in a third-party commercial publication (excluding your thesis/dissertation for which Amino acids play important roles in skin beautification. If you are the author of this article, you do not need to request permission to reproduce figuresĪnd diagrams provided correct acknowledgement is given. Peptides, including GHSs, are short chains of amino acids, which are little molecules that are the building blocks of peptides and proteins. The versatility of peptides as a building block is evident through the introduction of reactive units that allow for more robust, stiffer covalently (or chemically) crosslinked hydrogel networks, a necessity for 3D printing of mechanically stabilized materials, to be prepared. Provided correct acknowledgement is given. If you are an author contributing to an RSC publication, you do not need to request permission Please go to the Copyright Clearance Center request page. To request permission to reproduce material from this article in a commercial publication, Provided that the correct acknowledgement is given and it is not used for commercial purposes. To be brash, peptides are chains of amino acids bound together by peptide bonds, and when administered to the body they can have very great, and specific effects. This article in other publications, without requesting further permission from the RSC, Rapid and column-chromatography-free peptide chain elongation via a one-flow, three-component coupling approachĬreative Commons Attribution-NonCommercial 3.0 Unported Licence. These peptides are called carrier peptides and they help stabilize elements like manganese and copper to your skin. With this study, we achieved dramatic reductions in both time and cost compared with typical solid-phase synthesis. We also demonstrated a one-flow tripeptide synthesis via in situ preparation of α-NCA starting from three readily available protected amino acids. The total synthesis of beefy meaty peptide was achieved by repeating the 3CC approach with the addition of only one column chromatographic purification. 
We demonstrated the high-yielding and column-chromatography-free syntheses of 17 tripeptides, as well as a gram-scale synthesis of a tripeptide. 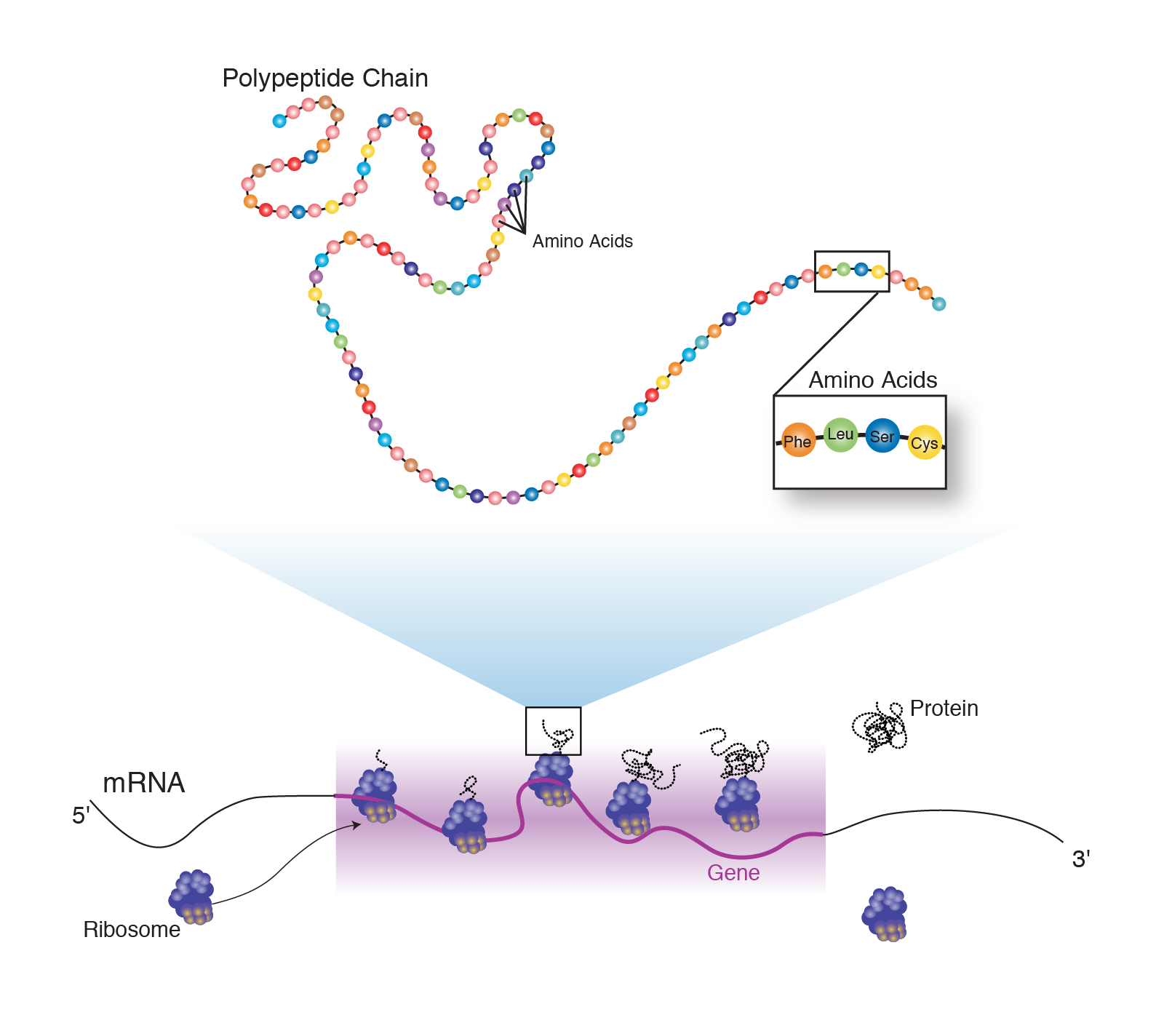
Here, we developed a rapid, mild, inexpensive, and column-chromatography-free peptide chain elongation via a one-flow, three-component coupling (3CC) approach that is the first to use α-amino acid N-carboxy anhydrides (α-NCAs) both as electrophiles and nucleophiles. Today, we know biology chooses just one of these chiral forms: DNA, RNA, and their building blocks are all right-handed. In 1848, French chemist Louis Pasteur discovered that some molecules essential for life exist in mirror image forms, much like our left and right hands. Both solid- and liquid-phase peptide syntheses suffer from a large number of synthetic steps, high cost, and/or tedious purification. Experiments suggest magnetic materials skewed early biomolecules. Short peptides are extremely important as drugs and building blocks for the syntheses of longer peptides.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
